Knowledge base
Expertise from A to H20
What are you looking for?

Membrane filtration
What is LOG reduction?

Microplastics
What are microplastics?
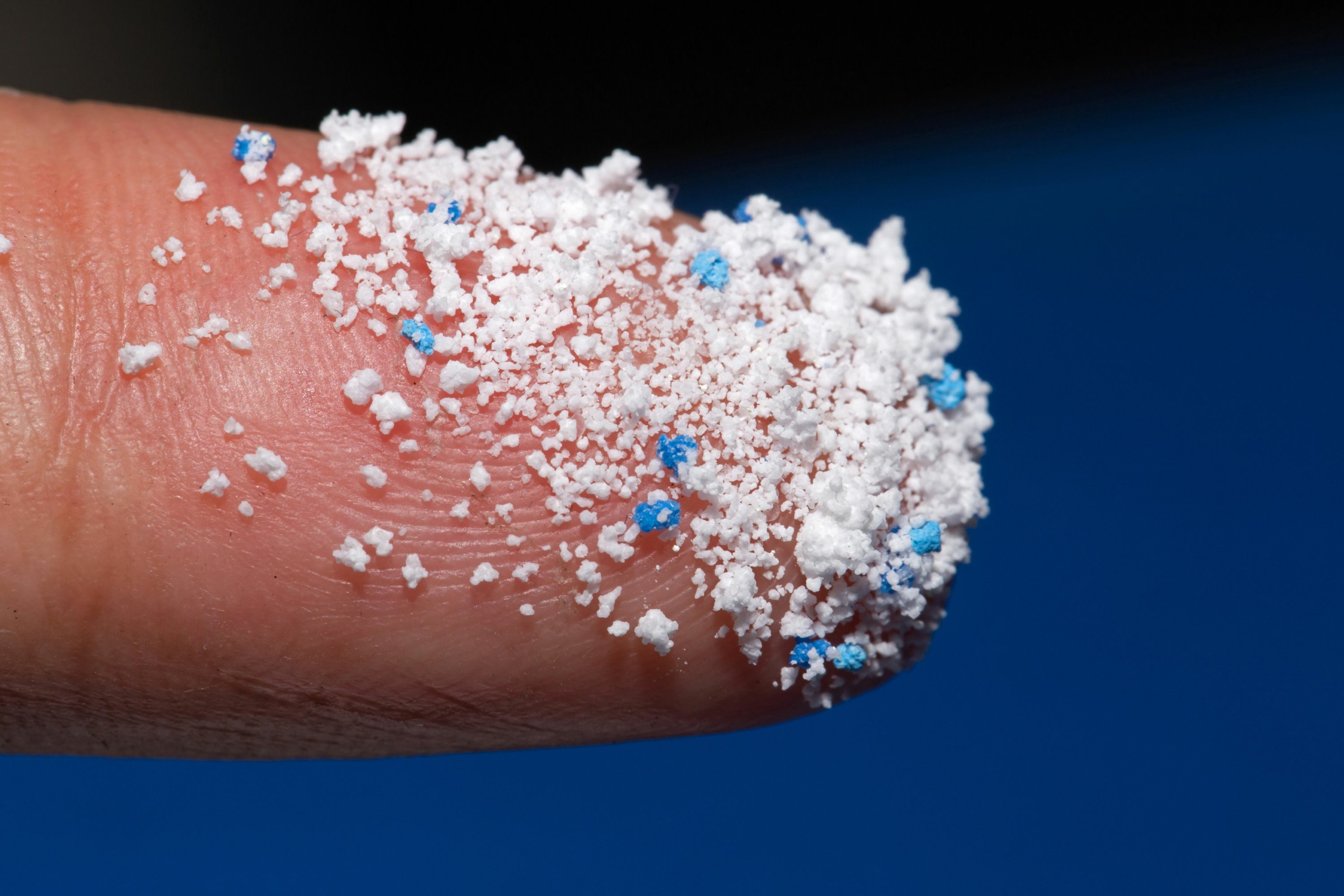
Microplastics
How does microplastic get into water?

Microplastics
How does a microplastic filter work?

Membrane filtration
What is ultrafiltration?

Membrane filtration
What is nanofiltration?

Membrane filtration
